By:Patrick Lukulay, PhD
Date:April 08, 2026
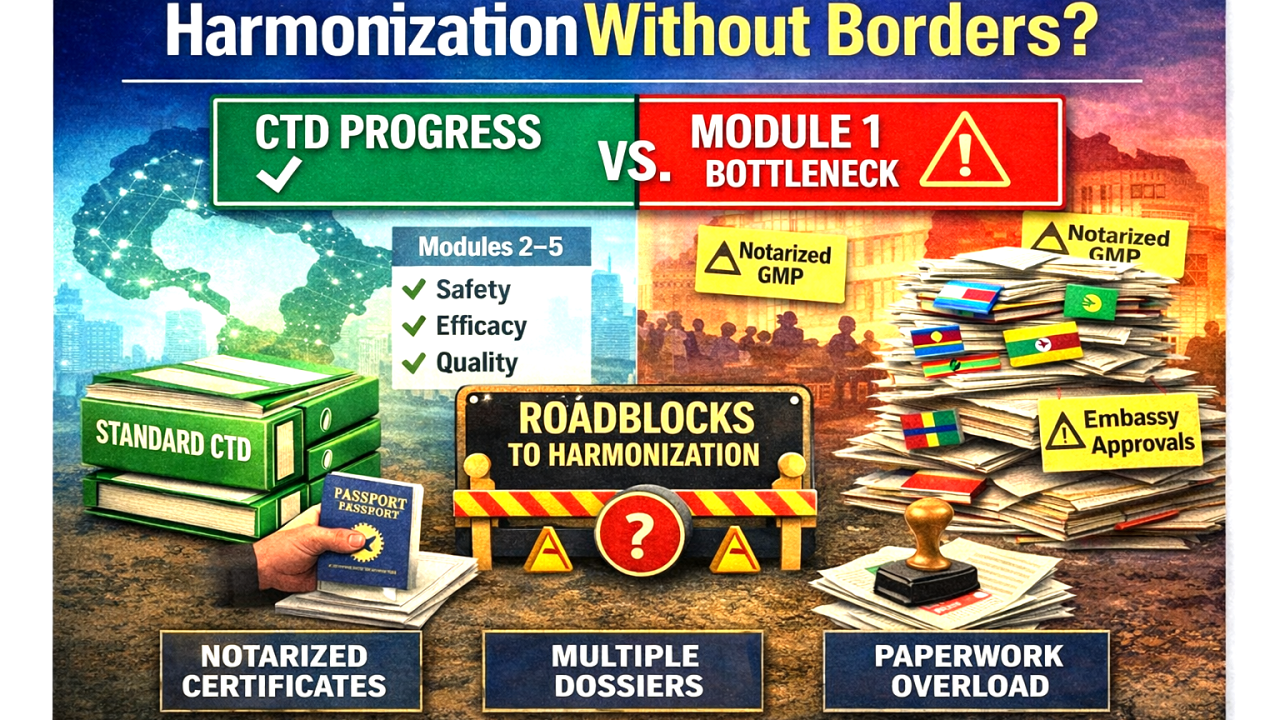
Unresolved bottlenecks in medicines regulatory harmonization in Africa.
Africa has crossed a critical threshold.
Thanks to the African Medicines Regulatory Harmonization (AMRH) initiative and the establishment of the African Medicines Agency (AMA), the adoption of the Common Technical Document (CTD) format has transformed how we evaluate the science of medicines across the continent.
For the first time, manufacturers are no longer reinventing the technical wheel for every country. That is real progress. That is worth celebrating.
But we now face an uncomfortable truth: We have harmonized the science (Modules 2–5) but left the administration (Module 1) in a state of fragmentation. And that fragmentation has become the single biggest bottleneck in registering products across African markets.
The Problem in Plain Sight
Module 1, the administrative gateway to any dossier, varies wildly from one country to the next. Consider what manufacturers are experiencing today:
- Duplicate data demands: One country requires extensive details on primary and secondary packaging as standalone administrative submissions, even though that exact information is already present in the Summary of Product Characteristics (SmPC). Imagine having to print different labels for different countries for the same product.
- The embassy chase: Some jurisdictions still demand physical notarization and embassy authentication of GMP certificates. Not because they distrust the issuing authority, but because processes have not caught up with reality. These same certificates can be verified in under 30 seconds on a competent regulatory authority’s website.
- Analogue in a digital age: We actively promote electronic submissions (e-CTD) and digital regulatory pathways. Yet we continue to require wet inks, stamps, and courier services for administrative proofs. This paradox is neither efficient nor defensible.
The Unintended Consequence of Partial Harmonization
The goal of harmonization was always speed, predictability, and access. But by leaving Module 1 largely uncoordinated, we have created a paradox:
Technical Dossier (CTD): One version.
Administrative Dossier (Module 1): potentially 54 versions.
Manufacturers are forced to prepare country-specific administrative packages that nullify the very benefits of harmonization promised. The result? Delayed timelines, duplicated effort, higher costs, and ultimately, slower patient access to essential medicines.
Yes, Local Context Matters. But Not Everything Needs to Be Different.
We fully recognize that certain country-specific requirements, such as pharmacovigilance language or local legal provisions cannot and should not be fully harmonized. National sovereignty and local public health priorities matter.
However, a significant portion of what is currently requested under Module 1 is standardizable, digitizable, and harmonizable.
Duplicating SmPC content into administrative checklists is not a public health necessity. Requiring embassy stamps for digitally verifiable certificates is not regulatory rigor. It is administrative friction.
A Call to Action for the African Medicines Agency (AMA)
We respectfully but urgently call on AMA, working with regional economic communities and National Regulatory Authorities (NRAs), to prioritize harmonization of Module 1 administrative requirements with the same rigor applied to technical Modules 2–5.
Concretely, this should include:
Standardized administrative templates and checklists across participating countries, with only limited, justified deviations.
Mutual recognition and digital verification of GMP certificates, replacing embassy authentication with real-time verification via competent authority websites or QR codes.
Full alignment with e-CTD and digital pathways, eliminating requirements for physical notarization where digital verification exists.
Without This, Harmonization Remains Incomplete
Africa has already proven that harmonization is possible. AMRH and AMA have laid a foundation worth defending.
But if manufacturers still need to prepare up to 54 different administrative dossiers, we have not truly achieved harmonization. We have simply standardized the science while ignoring the administrative part.
True harmonization is not just about aligning scientific standards. It is about removing every unnecessary barrier between a life-saving medicine and the patient who needs it.
The patient waiting in Lagos, Nairobi, or Johannesburg does not care about the elegance of our CTD. They care about access.
The author is Bio Usawa's Chief Operating Officer.
Bio Usawa will be at the American Society of Cataract and Refractive Surgery (ASCRS) Annual Meeting — uniting leaders advancing eye care through educati…
April 2026Bio Usawa at ADIS 2026: Connecting Science, Partnerships, and Impact
April 2026As part of Bio Usawa's engagement at the West Africa Biomanufacturing Regulation & Market Access Forum in Ghana last week, our Head of Global Regulatory…
March 2026